01
EMPOWERING MORE
FAMILIES, EARLIER
Genetron Health aims to radically improve the accessibility, affordability, and accuracy of early detection solutions for every family. Expanding from single cancer types to multiple cancer types, R&D breakthroughs and product commercialization must not only meet clinical needs, but also elevate service standards—only then can we reduce the mortality rate of cancer and foster lasting, generational impact.

02
LIVER CANCER, THE FIRST BREAKTHROUGH
In March 2019, Genetron Health—in collaboration with the National Cancer Center (PRC) and the Cancer Hospital of the China Academy of Medical Sciences—published research breakthroughs for a prospective cohort, concerning the early screening of liver cancer, in the Proceedings of the National Academy of Sciences (PNAS).
In August 2020, the Ministry of Science and Technology (PRC) launched a national R&D project for early cancer screening, selecting Genetron Health to participate in developing key technologies for the early detection of multiple cancer types.
In October 2021, Genetron
Health's original live cancer early screening model, M2P-HCC, was officially incorporated into the Screening and Surveillance of Hepatocellular Carcinoma in Patients with Chronic Hepatitis B Virus Infection published by the Chinese Journal of Hepatology.
In November 2021,Genetron Health’s liver cancer screening results and technology findings are included in the expert consensus on the role of hematological markers in the early clinical screening of hepatocellular carcinoma – submitted by the Infectious Disease Prevention and Control Division of the Chinese Preventive Medicine Association.
In August 2022, Genetron Health's early liver cancer screening technology was incorporated into the China Guideline for Liver Cancer Screening (2022, Beijing).
In December 2022, a research paper based on Genetron Health's early cancer screening technology Mutation Capsule Plus was published by Science Translational Medicine.



88%
SENSITIVITY
93%
SPECIFICITY
FINDING LIVER CANCER EARLY AT ASYMPTOMATIC STAGES
In March 2021, Genetron Health released new data results of a large, prospective cohort study on liquid-biopsy for liver cancer early detection. According to the data results, among a total of 1,615 HBsAg+ individuals, HCCscreen™️ achieved 88% sensitivity and 93% specificity, compared with 71% sensitivity and 95% specificity, respectively, by ultrasound plus alpha-fetoprotein (AFP) combined.

An exclusive strategic cooperation with CTTQ Pharma to jointly service the hospital market, providing precision oncology solutions for high-risk clinical patients with liver diseases.

A “Liver Cancer Early Screening Comprehensive Prevention Program” was launched in partnership with the National Cancer Center of China and the Huishan district government in Wuxi.
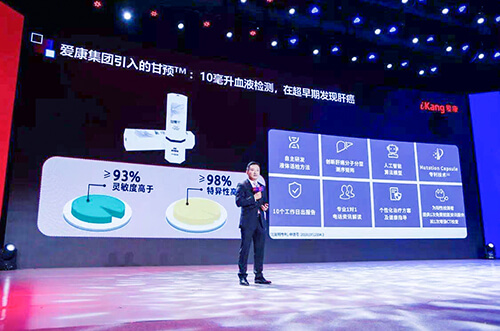
An exclusive strategic cooperation with iKang Group provides early screening products for the general population, covering over 150 locations throughout China.

A strategic cooperation with JD Health, a Chinese online healthcare platform, to build an"Innovative Internet model of cancer full-cycle management".